Feature Article

1 June 2026
Seven University of Auckland staff have been recognised in the King’s Birthday Honours, including Professor Elizabeth Rata who becomes a dame and Associate Professor Paul Baker who becomes a knight.
Sustainable impact
-

5 June 2026
Innovative solutions to environmental problems are urgently needed, because our wellbeing depends on it, says Professor Dan Tompkins, the new director of the Centre for Climate, Biodiversity and Society, Ngā Ara Whetū, at the University of Auckland.
-

3 June 2026
The first comprehensive guide to prison law in Aotearoa New Zealand is being developed with the support of a $153,000 grant.
-

3 June 2026
A timber building that kept itself centred through major shaking has aced a full‑scale earthquake test with no damage, with more than 60 industry professionals looking on.
Arts and culture
-

3 June 2026
Alumni profile: Violinist, organist and conductor Zosia Herlihy-O’Brien has found her calling bringing classical music to a wide range of audiences, including at London’s famed Royal Albert Hall.
-

3 June 2026
Vaiaso o le Gagana Sāmoa, Sāmoa Language Week in Aotearoa, is a time to celebrate those that uplift Pacific youth to succeed at Waipapa Taumata Rau.
-

2 June 2026
Alumni profile: Ruth Diver is experiencing wild success as a literary translator, including co-translating the English version of the bestselling Gisèle Pelicot memoir.
Business and economy
-

2 June 2026
Comment: Despite opposition from politicians on both the left and right, a form of mandatory saving for retirement could make economic sense, say Leonard Hong and Robert MacCulloch.
-
26 May 2026
Analysis: New Zealand needs stronger mechanisms to convert early promise into investable firms, says Rod McNaughton.
-

26 May 2026
Zolzaya Battsogt lost both her daughter and supervisor during her doctoral studies. This May, she graduated in their honour.
Education and society
-

28 May 2026
Comment: Education is complicated, and we should be wary of stories that suggest it is otherwise like the one the kids failing at maths, says Lisa Darragh.
-

28 May 2026
Celebrating Sāmoa Language Week, 31 May – 6 June. The journey of gagana Sāmoa is a lifelong one says Pro Vice-Chancellor Pacific, Fonoiā Professor Sipaea Jemaima Tiatia Siau.
-

27 May 2026
People living in deprived neighbourhoods have a significantly higher risk of premature ‘deaths of despair,' according to an international study using NZ data.
Te ao Māori
-

3 June 2026
Vaiaso o le Gagana Sāmoa, Sāmoa Language Week in Aotearoa, is a time to celebrate those that uplift Pacific youth to succeed at Waipapa Taumata Rau.
-

1 June 2026
Seven University of Auckland staff have been recognised in the King’s Birthday Honours, including Professor Elizabeth Rata who becomes a dame and Associate Professor Paul Baker who becomes a knight.
-

28 May 2026
The re-dedication of the Sir James Henare Research Centre recalls the layers of history and renews aspirations for the future.
Health and medicine
-
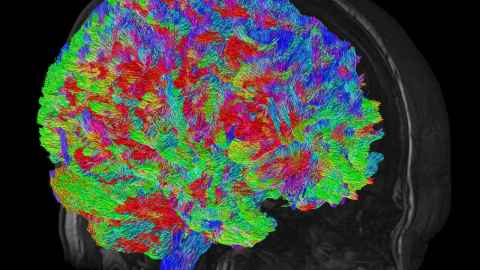
3 June 2026
Significant differences can be seen in the brains of recovering methamphetamine addicts, compared to those of non-users, a study involving University of Auckland scientists has found.
-

2 June 2026
Professor Dame Helen Danesh-Meyer's damehood recognises her significant contributions to ophthalmology.
-

2 June 2026
Hood Fellow Professor Russell Foster's visit has supported chronobiology research efforts in New Zealand and Sāmoa.
Politics and law
-

3 June 2026
The first comprehensive guide to prison law in Aotearoa New Zealand is being developed with the support of a $153,000 grant.
-

29 May 2026
With the can kicked down the road in terms of a referendum at the 2026 general election on extending our term to four years, the question remains: should we extend our parliamentary term beyond three years?
-

26 May 2026
Zolzaya Battsogt lost both her daughter and supervisor during her doctoral studies. This May, she graduated in their honour.
Science and technology
-

5 June 2026
Innovative solutions to environmental problems are urgently needed, because our wellbeing depends on it, says Professor Dan Tompkins, the new director of the Centre for Climate, Biodiversity and Society, Ngā Ara Whetū, at the University of Auckland.
-

3 June 2026
Feature: AI is developing at such breakneck speed it’s hard to know how the technology will next materially impact our lives and work. Anthony Doesburg asks alumni working on the front lines of tech about where AI is taking us in the next 12 months.
-

3 June 2026
A timber building that kept itself centred through major shaking has aced a full‑scale earthquake test with no damage, with more than 60 industry professionals looking on.
University news
-

1 June 2026
Seven University of Auckland staff have been recognised in the King’s Birthday Honours, including Professor Elizabeth Rata who becomes a dame and Associate Professor Paul Baker who becomes a knight.
-

25 May 2026
Josephine Davis will take over as Head of School of Nursing at the University’s Faculty of Medical and Health Sciences, marking a historic moment for Māori nursing leadership.
-

20 May 2026
A dawn ceremony on 19 May welcomed restored whakairo back to the James Henare Research Centre, reaffirming the centre’s place as a whare of mātauranga Māori.
The Challenge
-

28 May 2026
The re-dedication of the Sir James Henare Research Centre recalls the layers of history and renews aspirations for the future.
-

19 May 2026
The albatross population of Antipodes Island in the Southern Seas is critically endangered. With more data researchers hope to bring their numbers back.
-

2 March 2026
Piecing together how birds and humans interacted in the Marquesas Islands of French Polynesia hundreds of years ago is the focus of a revealing new study.
